Gasification
The idea of using wood as a fuel source to cut wood with a log splitter was very intriguing and somewhat comical. Alright, maybe completely comical and I just wanted to be able to say I cut wood with wood. Initially, using steam power was appealing and would have been a good segue to another back burner project of mine. However, the more I researched the dangers of high pressure and super heated steam it seemed like I would be playing with fire and decided I was not comfortable with it. Instead synGas generation was the alternative source to power a log splitter. So instead of playing with “fire” I opted to play with actual fire. Well, technically I would be delaying the fire and allowing a traditional internal combustion engine (ice) to consume the synGases in its combustion chamber.
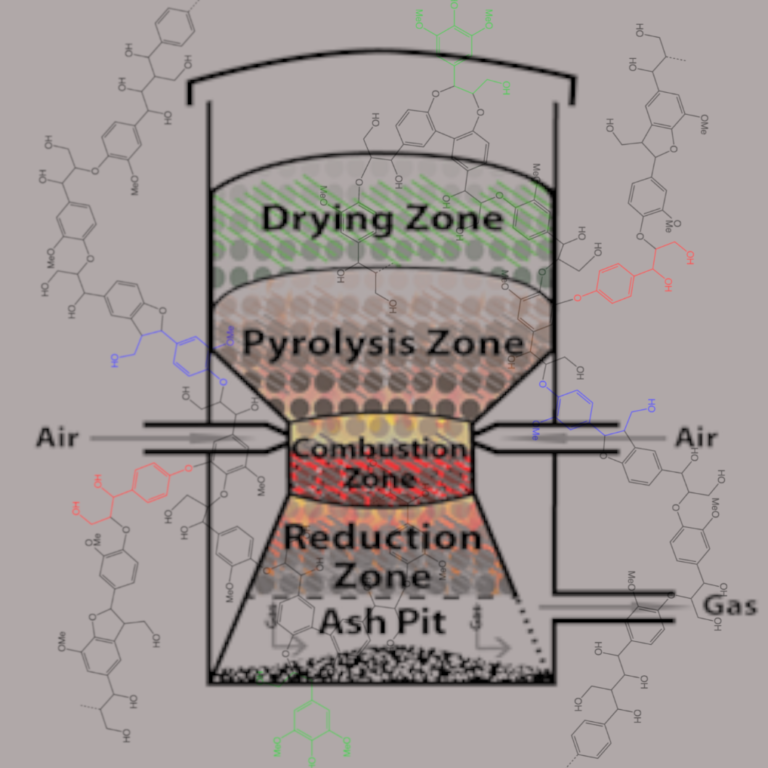
A little bit about combustion:
Wood and all fuel sources in a liquid or solid state do not actually burn. Instead, it is the gasses that are emitted through thermal decomposition that combust. The rapid oxidation of a fuel source (Fire) is only sustainable while the localized environment is able to reach a lower enthalpy state (or more stable chemical compound). As oxygen combusts with the vapors from the fuel the heat given off causes a feedback loop that allows more vapors to be emitted from the fuel source. The rapid oxidation process continues until one of the three edges of the fire triangle are removed.
The fire triangle describes what is required for a fire to ignite and continue the oxidation process. These three elements are: fuel, oxygen, and heat. If any of these elements are removed the oxidation process is no longer able to continue. Like all system, fire has an ideal ratio that optimizes the system. In this case: rapid oxidation. If you have ever used an acetylene torch you know that you have to set the oxygen and acetylene ratio to get optimal cutting. Too much fuel, or too much oxygen will have a negative effect on cutting performance. With that said, if you can adequately control part of the fire triangle you are able to postpone the oxidation process. Essentially, by limiting oxygen, what you are doing is breaking down the chemical structure of the fuel via super heating the fuel source. In the case of a gasifier you are causing thermal break down by only providing enough oxygen to sustain partial oxidation. This is meant to sustain heat within the combustion zone without completing the combustion reaction.
It should be noted that this process is very dangerous and highly volatile. One such danger can be illustrated by talking about flashovers that are often described in the context of a structure fire. Flashovers can be explosive. What happens is that there is plenty of heat and fuel in the localized environment however there is a depleted level of oxygen. The introduction of oxygen to the environment can restart the rapid oxidation process violently (see the video below). During the pyrolysis process you are creating the conditions necessary for a flashover to occur. While, each stage in the process has varying levels of risk based on the localized environment, all stages should be treated with great caution. Do your research if you are going to attempt this as this page is nothing more than an introduction to the topic.

Core concept:
Gasifiers (or pyrolitic distillation) have been used for several decades and its use tends to increase during times of increased instability of traditional petrofuels. The main objective of the gasification process is to superheat biomass material in a low oxygen environment to break the chemical bonds of the fuel source to produce synthesis gases (synGas). Essentially, you are creating the gas vapors that are needed for a fire but postponing the combustion of those gases until they are ignited within the combustion camber of a ICE or other heat motor. As described above, the low oxygen environment is critical because the synGases produced would complete the combustion reaction and the gasses obtained at the output of the gasifier would be in a more stable (lower enthalpy state) and would be of no use in creating mechanical energy.
The syngas is mainly composed of CO, H2, N2, CO2, and some hydrocarbons (CH4, C2H4, C2H6, etc.). Very small amounts of H2S, NH3, and tars may also be included
Source: https://www.sciencedirect.com/topics/earth-and-planetary-sciences/gasification#:
Some Drawbacks:
As you could imagine the traditional internal combustion engine is not easily adaptable to accept solid fuel sources like wood chips. Alternatively, the synGas produced from the gasification process is easily able to be fed in the intake of an internal combustion engine. While you do not have to worry about the atomization of synGases like you do with petrofuels, there are drawbacks to the use of a gasifier.
- Besides being volatile, SynGases are not safe to breath and can be very hazardous if care is not taken.
- The syngas production itself is fairly “clean” (free from foreign objects) however suspended soot is present as the gases exiting the thermal chamber. To remove the soot, the gases exiting the thermal chamber are directed to a separator that uses centrifugal forces to remove the soot and any other dense materials.
- The minimum temperature for generating producer gases is around 600-700°C. The energy of the gases need to be reduced significantly before entering the combustion chamber. Lowering synGas temperature results in greater energy density and can increase the working fluid capabilities of the gasses in the combustion chamber. Additionally, insufficient temperature reduction will likely exceed non-metal intake material ratings. In an effort to reduce the synGas temperatures an additional 55 gallon drum was introduce. This drum allows the gases to expand and pass over a scrubbing material. The syngas is then forced down two pvc expansion chambers.
- Unlike petrofuels there is an ash byproduct that needs to be periodically removed. While it a very clean burn compared to an open burn of biomass, a gasifier unit still needs to have this byproduct removed. This is because the thermal chamber must be air tight in order for the gasification process to work on account of needed to have tight control on the exposure to oxygen.
- Relative to petrofuels the maintenance required to power an engine with wood is very demanding. Instead of just filling up with 87 octane, with a gasifier you have to maintain a fuel source that is depleted much quicker do to its low energy density. Additionally, the moisture content is very important. Because you need to thermally decompose the fuel you also have to remove all moisture in the fuel. This is built into the gasification design however the process of drying is effectively limiting the heat leg of the fire triangle and requires that more combustion happen within the gasifier for the drying process. Ultimately this leads to less available synGas within the ICE combustion chamber.
The unit in the image below was used to connect directly to an traditional internal combustion engine that was powering a log splitter. Unfortunately, I am unable to locate the video of the engine running off the synthesis gases produced by this gasifier.

